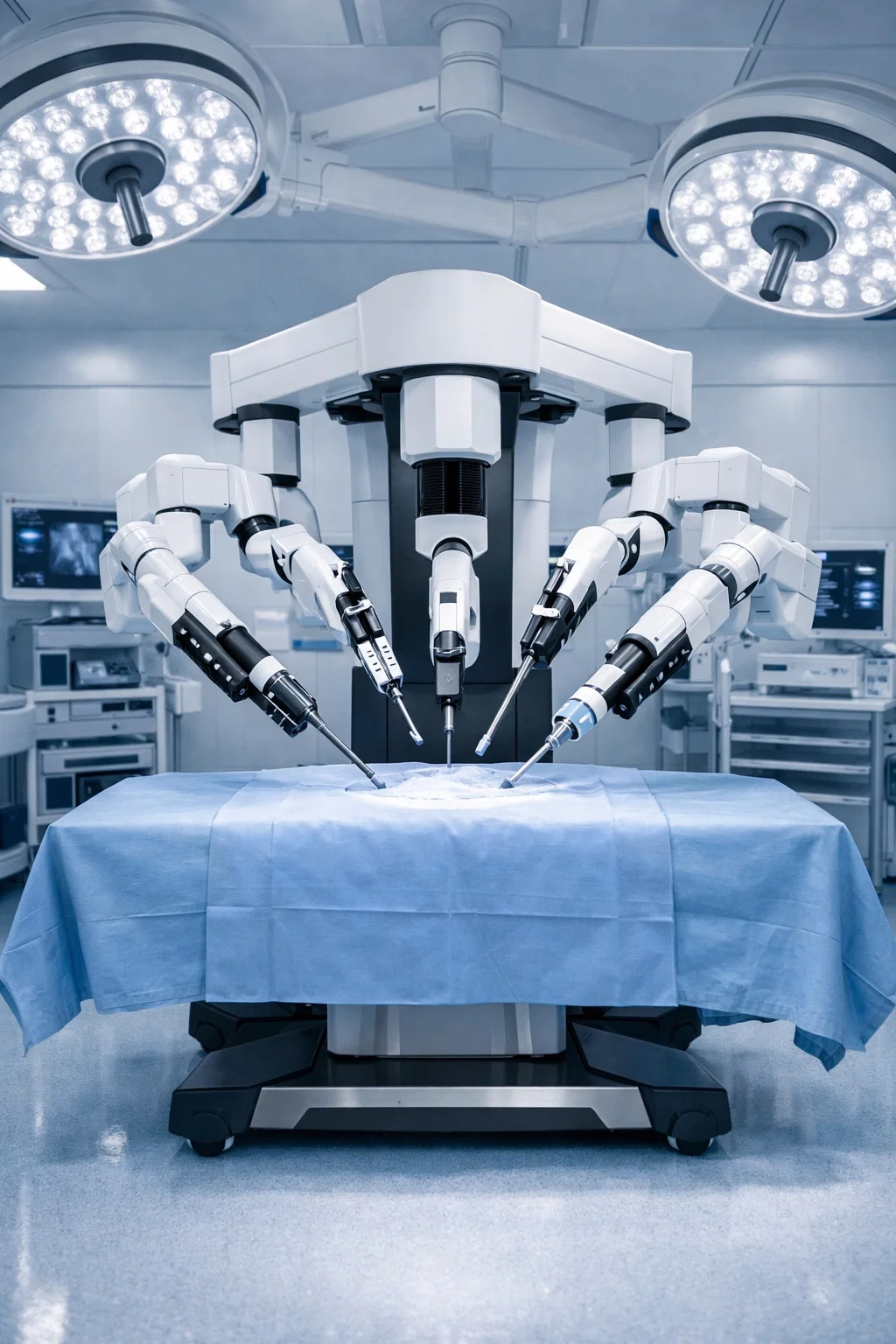
Rocuvex Medical was founded with a clear mission:
…to help surgical programs maintain clinical excellence while addressing the growing financial pressures of robotic surgery. With over 12 years of experience spanning robotic capital equipment, clinical environments, and operating room workflows, our leadership has seen firsthand the increasing cost burden associated with Da Vinci Dv5 & Xi robotic instruments.
As utilization continues to rise, so does the strain on hospital budgets and operating margins. Rocuvex Medical was established to bridge that gap, serving as a trusted distributor that provides hospitals with access to FDA-cleared remanufactured robotic instruments and certified pre-owned inventory through leading, approved manufacturing partners.
Today, Rocuvex delivers these solutions through a nationwide model built on dedicated, localized support, ensuring hospitals across the United States receive hands-on service, seamless program implementation, and reliable access to cost-saving robotic instrument solutions.


Providing Access to Fully Reprocessed Da Vinci Dv5 & Xi Robotic Instruments
We specialize in providing hospitals with access to fully reprocessed Da Vinci Dv5 & Xi robotic instruments that meet rigorous quality and compliance standards, sourced through trusted, FDA-cleared manufacturing partners. Our solutions integrate seamlessly into existing surgical workflows, helping reduce costs without compromising performance, safety, or surgeon confidence.
20–25% reduction in DaVinci instrument costs
Reliable turnaround times with simplified ordering
Dedicated regional support and direct accountability
FAQ
Frequently Asked Questions
Find answers to the most common questions about our remanufactured instruments, FDA clearance, and cost savings program.
What is remanufactured robotic instrument?
A remanufactured instrument is a used robotic surgical instrument that has been restored through an FDA-cleared process, tested for performance, and made available for reuse at a lower cost.
Is the remanufacturing process FDA approved?
Yes. Restore Robotics holds the industry's only FDA 510(k) clearance for remanufacturing Da Vinci Xi robotic instruments.
Which robotic systems are compatible?
Our instruments are compatible with Da Vinci Xi and DV5 robotic surgical systems.
How much can our hospital save?
Hospitals typically save 20%+ on remanufactured instruments and 25%+ per use on certified pre-owned inventory compared to new OEM instruments.
Are remanufactured instruments as safe as new ones?
Yes. Every instrument undergoes visual inspection, functional performance testing, electrical verification, and quality control validation before use.
Who provides the warranty?
Restore Robotics becomes the manufacturer of record and provides full warranty coverage for all remanufactured instruments.
How does the collection program work?
Used instruments are placed in provided collection trays, secured in reusable shipping containers, and sent to the remanufacturing facility for inspection and processing. Secured and reusable shipping containers are provided by Encore Medical Device Repair for free and sent to our partners at Restore Robotics remanufacturing facility for inspection and processing.
Does the program disrupt our surgical workflow?
No. The program is designed to integrate seamlessly into existing hospital processes with minimal disruption to surgical operations.
What areas do you serve?
Rocuvex Medical serves hospitals and surgical centers nationwide, providing dedicated local support across the United States.
How do we get started?
Contact our team to request a complimentary savings analysis. We will evaluate your instrument usage and estimate your potential cost savings.
Request a savings analysis for your hospital
Every robotic surgery program is different. Contact our team to estimate your potential cost savings with remanufactured and certified pre-owned instruments.